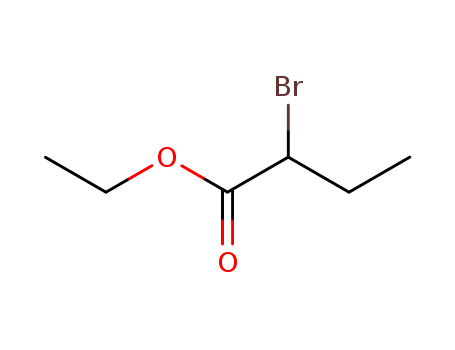
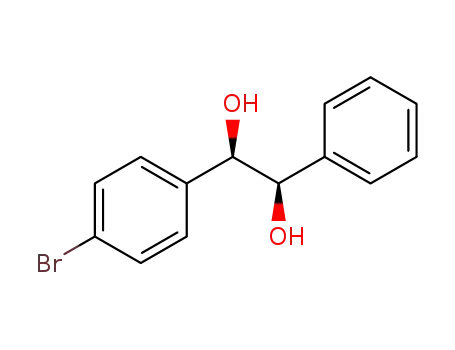
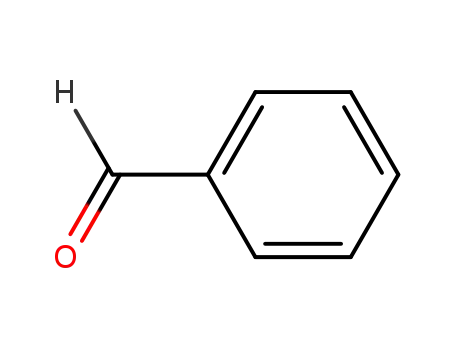
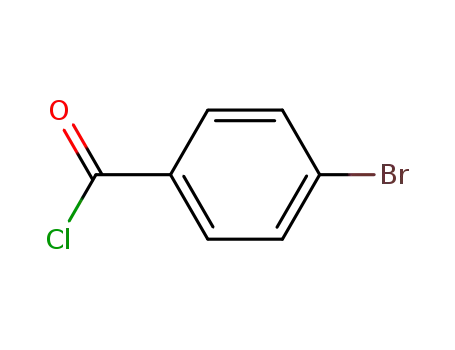
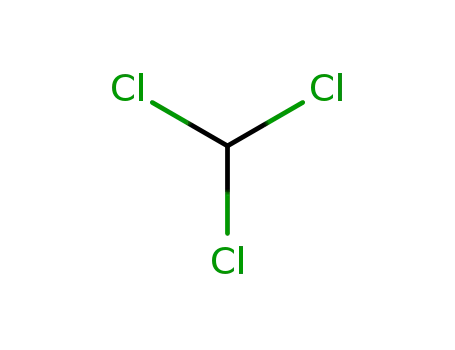
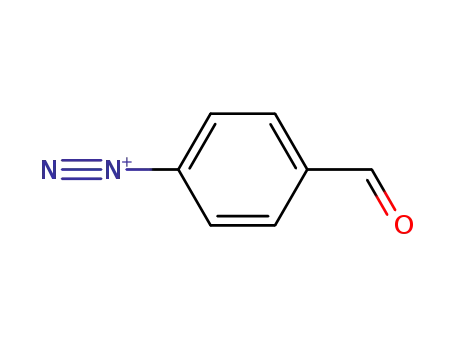
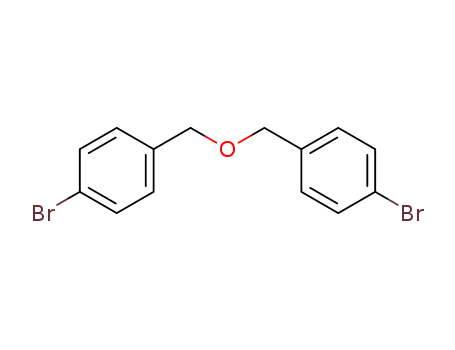
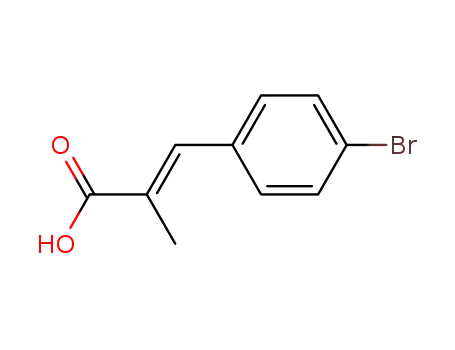
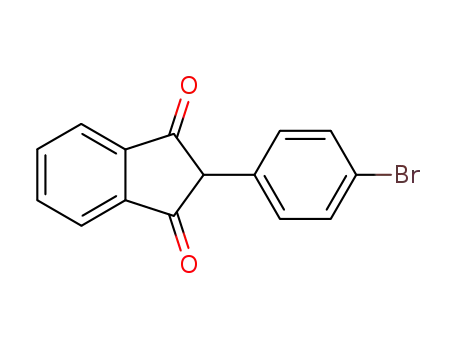
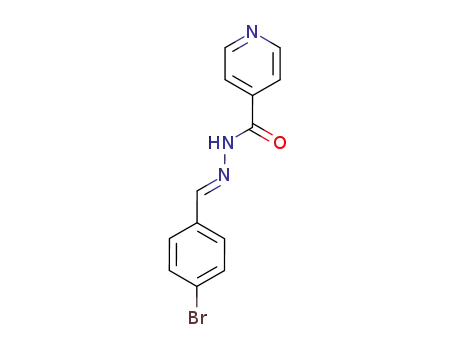
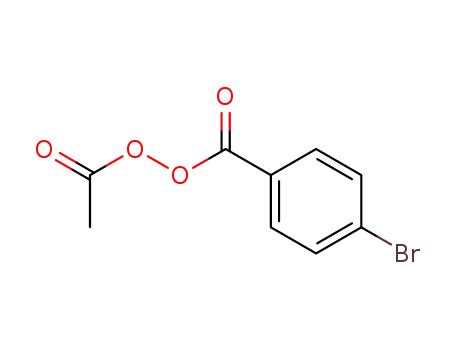
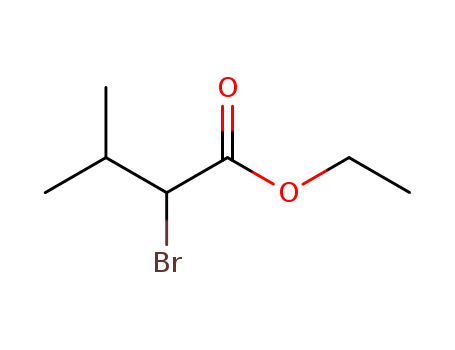
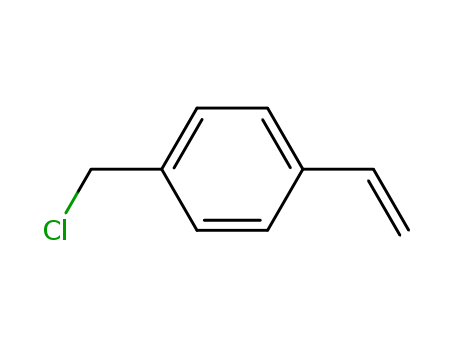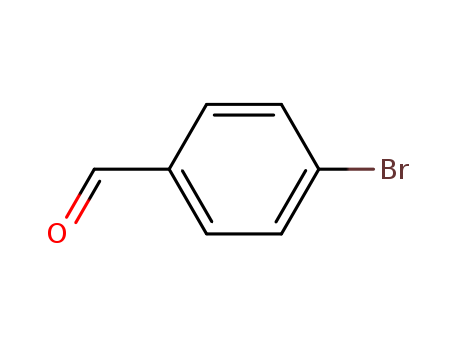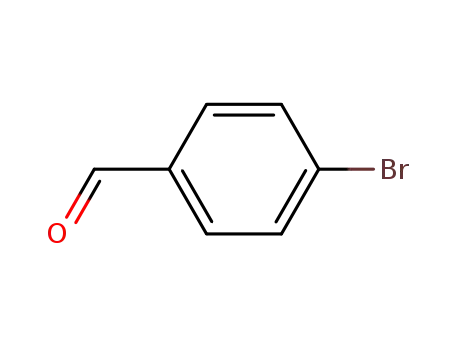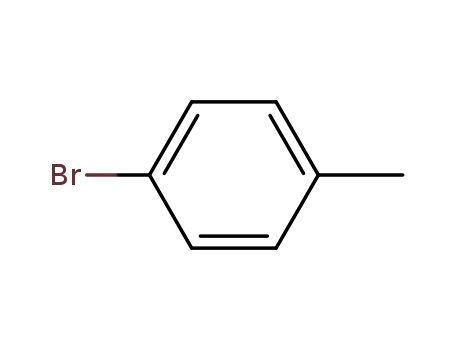Regioselective radical aminofluorination of styrenes
Zhang, Hongwei,Song, Yongcheng,Zhao, Jinbo,Zhang, Jingping,Zhang, Qian
, p. 11079 - 11083 (2014)
The copper-catalyzed radical aminofluori...
Microwave-assisted chemoselective regeneration of carbonyl compounds from oximes by silica chromate/WET SiO2 under solvent-free conditions
Zolfigol, Mohammad Ali,Khazaei, Ardeshir,Ghorbani-Choghamarani, Arash,Rostami, Amin
, p. 2453 - 2458 (2006)
The regeneration of carbonyl compounds f...
A mild and efficient method for the oxidation of benzylic alcohols by two-phase electrolysis
Raju, Thasan,Manivasagan, Sankar,Revathy, Balachandran,Kulangiappar, Kumarasamy,Muthukumaran, Arunachalam
, p. 3681 - 3684 (2007)
Electrochemical oxidation of benzylic an...
Novel Schiff base Mn(III) and Co(II) complexes supported on Co nanoparticles: Efficient and recyclable magnetic nanocatalysts for alcohol oxidation
Keypour, Hassan,Saremi, Shokoufeh Ghahri,Veisi, Hojat,Azadbakht, Reza
, p. 77020 - 77029 (2016)
In this study, efficient and highly sele...
Zinc Bismuthate Zn(BiO3)2. I. A Useful Oxidizing Agent for the Efficient Oxidation of Organic Compounds
Firouzabadi, Habib,Mohammadpour-Baltork, Iraj
, p. 1131 - 1134 (1992)
Zinc bismuthate is an easily prepared an...
-
Trahanovsky et al.
, p. 3865 (1967)
-
-
Huggins,Yokley
, p. 1160 (1942)
-
Chromium(0)-carbene complexes as carbene sources: Self-dimerization and inter- and intramolecular C-H insertion reactions catalyzed by Pd(OAc)2
Sierra,Mancheno,Saez,Del Amo
, p. 6812 - 6813 (1998)
-
Cerium-Complex-Catalyzed Oxidation of Arylmethanols under Atmospheric Pressure of Dioxygen and Its Mechanism through a Side-On μ-Peroxo Dicerium(IV) Complex
Paul, Mitali,Shirase, Satoru,Morimoto, Yuma,Mathey, Laurent,Murugesapandian, Balasubramanian,Tanaka, Shinji,Itoh, Shinobu,Tsurugi, Hayato,Mashima, Kazushi
, p. 4008 - 4014 (2016)
A new CeIV complex [Ce{NH(CH2CH2N=CHC6H2...
Mercury(II) Oxide/35percent Aqueous Tetrafluoroboric Acid/Tetrahydrofuran: An Improved Reagent for the Hydrolysis of Cyclic Hemithioacetals, Dithioacetals, and Ortho Esters
Degani, Iacopo,Fochi, Rita,Regondi, Valeria
, p. 51 - 53 (1981)
-
Synthetic utility of iodic acid in the oxidation of benzylic alcohols to aromatic aldehydes and ketones
Imai, Sho,Togo, Hideo
, p. 6948 - 6954 (2016)
Various primary and secondary benzylic a...
Oxidations with IBX: Benzyl halides to carbonyl compounds, and the one-pot conversion of olefins to 1,2-diketones
Moorthy, Jarugu Narasimha,Singhal, Nidhi,Senapati, Kalyan
, p. 1757 - 1761 (2006)
A variety of benzyl halides were convert...
gem-Difluorination of 2,2-diaryl-1,3-dithiolanes by Selectfluor and pyridinium polyhydrogen fluoride
Reddy, V. Prakash,Alleti, Ramesh,Perambuduru, Meher K.,Welz-Biermann, Urs,Buchholz, Herwig,Prakash, G. K. Surya
, p. 654 - 656 (2005)
2,2-Diaryl-1,3-dithiolanes, readily obta...
Selective oxidation at carbon adjacent to aromatic systems with IBX
Nicolaou,Baran,Zhong
, p. 3183 - 3185 (2001)
-
"Click Chemistry" Mediated Functional Microporous Organic Nanotube Networks for Heterogeneous Catalysis
Yu, Wei,Zhou, Minghong,Wang, Tianqi,He, Zidong,Shi, Buyin,Xu, Yang,Huang, Kun
, p. 5776 - 5779 (2017)
The synthesis of azide functional microp...
Pd(0)-catalyzed PMHS reductions of aromatic acid chlorides to aldehydes
Lee, Kyoungsoo,Maleczka Jr., Robert E.
, p. 1887 - 1888 (2006)
Contrary to previous reports, polymethyl...
Competing bromination and oxidation pathways in acid bromate solutions: An experimental and theoretical study
Rothenberg, Gadi,Beadnall, Richard M.H.,McGrady, John E.,Clark, James H.
, p. 630 - 635 (2002)
The qualities that render concentrated H...
Tributylmagnesium ate complex-mediated novel bromine-magnesium exchange reaction for selective monosubstitution of dibromoarenes
Iida, Takehiko,Wada, Toshihiro,Tomimoto, Koji,Mase, Toshiaki
, p. 4841 - 4844 (2001)
Lithium tributylmagnesate complex (n-Bu3...
{-N+(CH3)2CH2CH 2N+[(CH3)2](CH2) 4-}n S2O82-: A selective and recyclable reagent for oxidation of alcohols and hydroquinones in water
Tajbakhsh, Mahmood,Lakouraj, Moslem Mansour,Ramzanian-Lehmali, Farhad
, p. 1724 - 1728 (2006)
This article describes the oxidation of ...
Preparation, characterization and application of sulfated Ti-SBA-15 catalyst for oxidation of benzyl alcohol to benzaldehyde
Sharma, Rajesh V.,Soni, Kapil K.,Dalai, Ajay K.
, p. 87 - 91 (2012)
Benzaldehyde is an important chemical an...
Mesityltriphenylbismuthonium tetrafluoroborate as an efficient bismuth(V) oxidant: remarkable steric effects on reaction rates and chemoselectivities in alcohol oxidation
Matano, Yoshihiro,Suzuki, Takeshi,Shinokura, Tomonori,Imahori, Hiroshi
, p. 2885 - 2888 (2007)
In the presence of N,N,N′,N′-tetramethyl...
BIOSYNTHESIS OF AZETIDINE-2-CARBOXYLIC ACID IN CONVALLARIA MAJALIS: STUDIES WITH N-15 LABELLED PRECURSORS
Leete, Edward,Louters, Laurence L.,Rao, H. S. Prakash
, p. 2753 - 2758 (1986)
Key Word Index - Convallaria majalis; Li...
Mild oxidation of benzylic amines into aldehydes using an oxidative Polonovski-like process
Desjardins, Samuel,Jacquemot, Guillaume,Canesi, Sylvain
, p. 1497 - 1500 (2012)
A chemoselective and environmentally ben...
AIBN/NaBr-promoted aerobic oxidation of benzylic alcohols via a radical process
Liu, Chengkou,Fang, Zheng,Yang, Zhao,Li, Qingwen,Guo, Shiyu,Guo, Kai
, p. 577 - 581 (2015)
An economic and practical AIBN-initiated...
Rapid and selective oxidation of benzyl alcohols to aldehydes and ketones with novel vanadium polyoxometalate under solvent-free conditions
Dewan, Anindita,Sarma, Tridib,Bora, Utpal,Kakati, Dilip K.
, p. 2563 - 2565 (2011)
A novel vanadium polyoxometalate [(C6H5C...
Facile oxidation of alcohols to carbonyl compounds using a tris(2-methylphenyl)-bismuth dichloride-DBU binary system
Matano, Yoshihiro,Nomura, Hazumi
, p. 3028 - 3031 (2002)
High efficiency and chemoselectivity as ...
Controlled reduction of activated primary and secondary amides into aldehydes with diisobutylaluminum hydride
Azeez, Sadaf,Kandasamy, Jeyakumar,Sabiah, Shahulhameed,Sureshbabu, Popuri
supporting information, p. 2048 - 2053 (2022/03/31)
A practical method is disclosed for the ...
Highly atom efficient synthesis of 2,2,4,5-tetrasubstituted 3(2H)-furanones having both hydroxyl and amino substituents
Antony, Jesna,Mathai, Sindhu,Natarajan, Rakesh,P. Musthafa, Sumi,Rappai, John P.,S. Devaky, Karakkattu
supporting information, (2022/02/25)
We have developed a highly atom efficien...
Poly-N-bromosulfonamide-melamine as a novel brominating reagent for regioselective ipso-bromination of arylboronic acids
Alavinia, Sedigheh,Ghorbani-Vaghei, Ramin
, p. 1269 - 1276 (2021/08/27)
A practical synthetic method for the syn...
Poly(ethylene glycol) dimethyl ether mediated oxidative scission of aromatic olefins to carbonyl compounds by molecular oxygen
Yu, Tao,Guo, Mingqing,Wen, Simiaomiao,Zhao, Rongrong,Wang, Jinlong,Sun, Yanli,Liu, Qixing,Zhou, Haifeng
, p. 13848 - 13852 (2021/04/22)
A simple, and practical oxidative scissi...
 English
English 中文
中文
 English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego