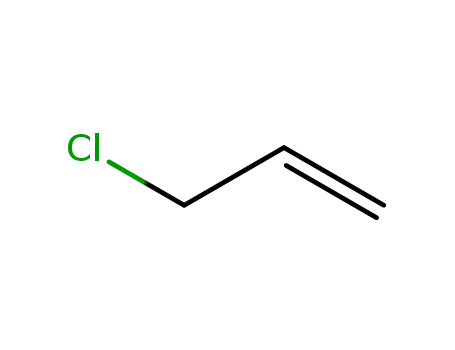|
Production Methods
|
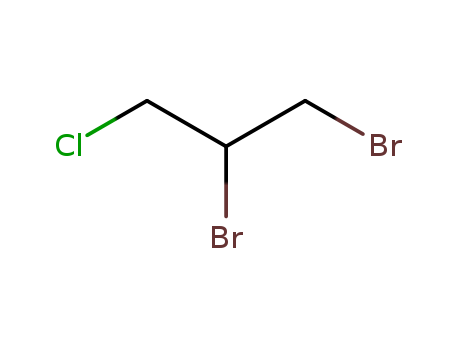
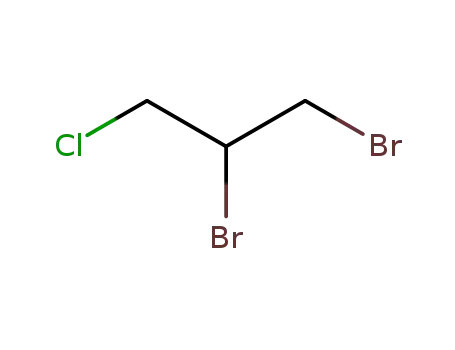
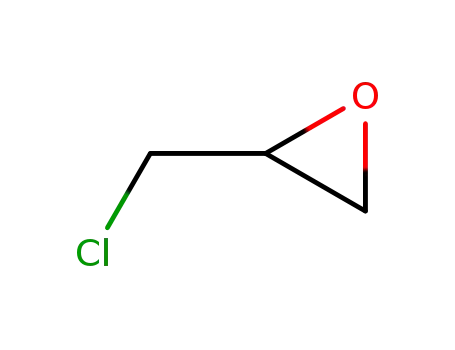
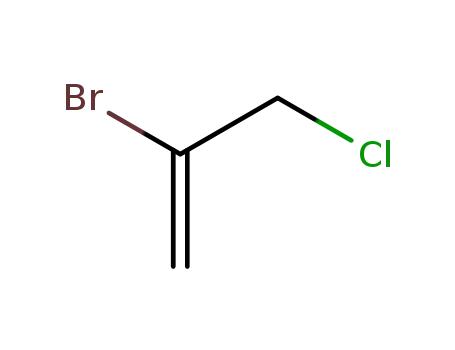
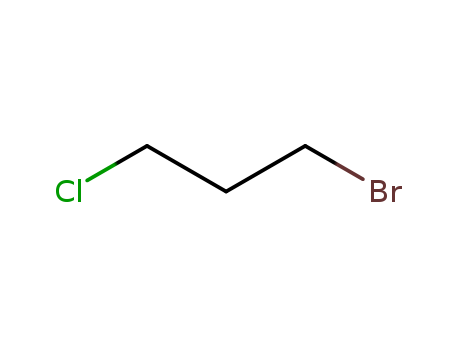
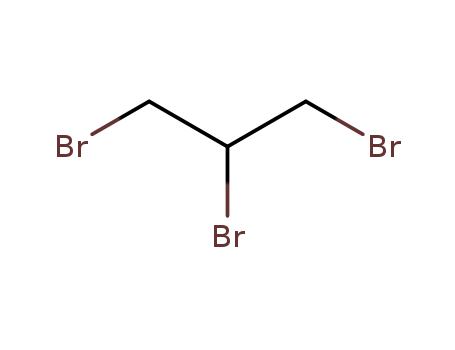
DBCP is produced by liquid phase addition of bromine to
allyl chloride. It was first produced commercially in the
United States in 1955. |
|
Air & Water Reactions
|
Flammable. Soluble in water. Hydrolyzed in alkali. |
|
Reactivity Profile
|
1,2-Dibromo-3-chloropropane reacts with chemically active metals such as aluminum, magnesium, tin and their alloys. 1,2-Dibromo-3-chloropropane will attack some rubber materials and coatings. |
|
Trade name
|
BBC 12?; FUMAGONE?;
FUMAZONE?[C]; MEMATOCIDE?; NEMABROM?;
NEMAFUM?; NEMAGON?[C]; NEMAGON SOIL
FUMIGANT?[C]; NEMANAX?; NEMAPAZ?;
NEMASET?; NEMATOCIDE?[C]; NEMATOX?;
NEMAZON?; OS 1897?; OXY BCP?[C]; SD 1897? |
|
Safety Profile
|
Confirmed human
carcinogen with experimental carcinogenic
and teratogenic data. Poison by ingestion,
inhalation, and subcutaneous routes.
Moderately toxic by skin contact. An eye
and severe skin irritant. Narcotic in high
concentrations. Has been implicated in
causing human sterihty in male factory
workers. Human mutation data reported. A
soil fumigant. Combustible. When heated to
decomposition it emits toxic fumes of Cl
and Br-. See also CHLORIDES and
BROLVIDES. |
|
Carcinogenicity
|
1,2-Dibromo-3-chloropropane is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity fromstudies in experimental animals. |
|
Environmental Fate
|
Biological. Biodegradation is not expected to be significant in removing 1,2-dibromo-
3-chloropropane. In aerobic soil columns, no degradation was observed after 25 days
(Wilson et al., 1981a).
Soil. Soil water cultures converted 1,2-dibromo-3-chloropropane to n-propanol, bromide
and chloride ions. Precursors to the alcohol formation include allyl chloride and allyl
alcohol (Castro and Belser, 1968). The reported half-life in soil is 6 months (Jury et al.,
1987).
Groundwater. According to the U.S. EPA (1986) 1,2-dibromo-3-chloropropane has a
high potential to leach to groundwater.
Chemical/Physical. 1,2-Dibromo-3-chloropropane is subject to both neutral and basemediate
hydrolysis (Kollig, 1993). Under neutral conditions, the chlorine or bromine atoms
may be displaced by hydroxyl ions. If nucleophilic attack occurs at the carbon-chlorine
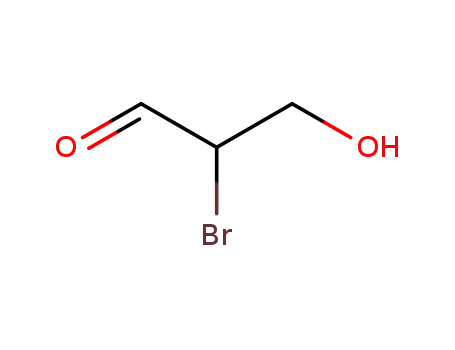
bond, 2,3-dibromopropanol is formed which reacts further to give 2,3-dihydroxybromopropane
via the intermediate epibromohydrin. 2,3-Dihydroxybromopropane will undergo
hydrolysis via the intermediate 1-hydroxy-2,3-propylene oxide which further reacts with
water to give glycerol. If the nucleophilic attack occurs at the carbon-bromine bond, 2-
bromo-3-chloropropanol is formed which further reacts forming the end product glycerol
(Kollig, 1993). If hydrolysis of 1,2-dibromo-2-chloropropane occurs under basic conditions,
the compound will undergo dehydrohalogenation to form 2-bromo-3-chloropropene
and 2,3-dibromo-1-propene as intermediates. Both compounds are subject to further attack
forming 2-bromo-3-hydroxypropene as the end product (Burlinson et al., 1982; Kollig,
1993). The hydrolysis half-life at pH 7 and 25°C was calculated to be 38 years (Burlinson
et al., 1982; Ellington et al., 1986).
Emits toxic chloride and bromide fumes when heated to decomposition (Lewis, 1990). |
|
Toxicity evaluation
|
DBCP in soil can volatilize from near-surface soil. Based on
estimated Koc values of 149 in Lincoln fine sand and 128 in an
unspecified soil, DBCP will be highly mobile in soil and has the
capacity to leach into groundwater. It is estimated that the
volatilization half-life for this chemical will vary between 0.6
days in dry soil with low soil organic content and 26.2 days in
a wet soil containing high values of soil organic content. It is
important to notice that fields treated with DBCP will present
a significant retardation in the volatilization loss from the soil
if plastic coverings are used.DBCP will exist primarily in the vapor phase in the
ambient atmosphere. It is expected that very small amounts of
this chemical will be present in the particulate phase, and this
could explain the fact that dry deposition to the earth’s surface
will not result in a significant removal process. DBCP presents
high water solubility, and then small amounts present in the air
can be removed by wet deposition.If found in water, DBCP will volatilize quite rapidly to the
atmosphere; the estimated volatilization half-life from a pond
is 8 days. Using again the estimated Koc values of 128–149, this
chemical is not expected to adsorb significantly to sediment
and suspended organic matter. Based on an estimated bioconcentration
factor (BCF) of 11.2, it is not expected to bioconcentrate
in fish and other aquatic organisms. There is no
sufficient data to indicate that DBCP could be a potential risk in
terms of biomagnifications in different aquatic or terrestrial
food chains. However, some biodegradation of DBCP in
groundwater might ensue under anaerobic conditions. |
|
Physical properties
|
Colorless when pure, however, technical grades are yellowish to dark brown. Pungent odor at high
concentrations |
|
General Description
|
A colorless liquid. Denser than water. Flash point 170°F. Boiling point 195°F. Toxic by ingestion and inhalation. Used as a pesticide and fumigant. |
|
Agricultural Uses
|
Nematicide, Fumigant: DBCP has been used in agriculture as a nematicide
since 1955, being supplied for such use in the forms of liquid
concentrate, emulsifiable concentrate, powder, granules,
and solid material. A rebuttable presumption against
registration for pesticide uses was issued by U.S. EPA on
September 22, 1977, on the basis of oncogenicity and reproductive
effects. Then, as of November 3, 1977, EPA in a
further action suspended all registrations of end-use products,
subject to various specific restrictions. Not listed as
registered in EU countries. |
 English
English 中文
中文
 English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego
English
Japanese
Russian
Korean
गोंगेन हें नांव
Deutsch
Corsu
Guarani
Hausa
Cymraeg
Nederlands
Aymara
Français
Kreyòl ayisyen
čeština
ʻŌlelo Hawaiʻi
डोग्रिड ने दी
ภาษาไทย
հայերեն
فارسی
Hmoob
ދިވެހި
भोजपुरी
繁體中文
Türkçe
हिंदी
беларускі
български
tur
Gaeilge
ગુજરાતી
Magyar
Eesti keel
بالعربية
বাংলা
Azərbaycan
Português
Suid-Afrikaanse Dutch taal
کوردی-سۆرانی
Ελληνικά
español
Frysk
dansk
አማርኛ
Bamanankan
euskara
Italiano
Tiếng Việt
অসমীয়া
català
Suomalainen
Eʋegbe
Hrvatski
Cebuano
Gàidhlig na h-Alba
bosanski
galego